Acetylenedicarboxylic acid
MOQ : 5 Grams
Acetylenedicarboxylic acid Specification
- Shelf Life
- 24 months from date of manufacturing
- Density
- 1.613 Gram per cubic centimeter(g/cm3)
- Physical State
- Solid
- Molecular Weight
- 130.07 g/mol
- Melting Point
- >300C (decomposes)
- Flash Point
- Not flammable
- Size
- Custom packaging available
- Storage Instructions
- Keep container tightly closed in a dry and well-ventilated place
- Packaging Type
- Bottle
- Purity
- 99%
- Usage
- For lab use only, not for drug, household or other uses
- Molecular Formula
- C4H2O4
- Boiling point
- Not applicable (decomposes before boiling)
- CAS No
- 142-45-0
- Grade
- Analytical Grade
- Type
- University Lab Chemicals
- Application
- Research, Laboratory usage, Organic synthesis
- Appearance
- White crystalline powder
- Purity(%)
- 99%
- Solubility
- Soluble in water
- Handling Precautions
- Wear suitable protective clothing, gloves and eye/face protection
- Moisture Content
- < 0.5%
- EC Number
- 205-528-1
- HS Code
- 29171990
- Hazard Class
- Not classified as hazardous under GHS
- Odor
- Odorless
Acetylenedicarboxylic acid Trade Information
- Minimum Order Quantity
- 5 Grams
- FOB Port
- Nhava- sheva, India
- Payment Terms
- Cheque, Cash Advance (CA)
- Supply Ability
- Gram
- Delivery Time
- 1 Week
- Sample Available
- Yes
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Main Export Market(s)
- Western Europe, North America, Eastern Europe, Middle East, Central America, Africa, South America, Australia, Asia
- Main Domestic Market
- All India
- Certifications
- ISO 14001 : 2015 ISO 9001 : 2008 OHSAS 18001 : 2007 WHO GMP
About Acetylenedicarboxylic acid
LC191 Acetylenedicarboxylic acidfor synthesis
| Order number | Packaging | Quantity | Price |
| AC27191 | Glass bottle | 10 g | 27.675 |
| Product information | |
| Synonyms | Butynedioic acid |
| Hill Formula | C4H2O4 |
| HS Code | 2917 19 90 |
| EC number | 205-536-0 |
| Molar mass | 114.05 g/mol |
| Storage | Store at +2C to +8C. |
| CAS number | 142-45-0 |
| Chemical and physical data | |
| Solubility | (20 C) soluble |
| Melting point | 180 - 185 C |
| Molar mass | 114.05 g/mol |
| Bulk density | 550 kg/m3 |
| Flash point | 131 C |
| Safety information | |
| Storage class | 10 - 13 Other liquids and solids |
| WGK | WGK 3 highly water endangering |
| Disposal | 4 Liquid organic acids or solutions should be diluted, if necessary, and carefully neutralized with sodium hydrogen carbonate (Cat. No. 106323) or with sodium hydroxide (Cat. No 106462). Before filling into container D, check the pH with pH universal indicator strips (Cat. No. 109535). |
| Transport information | |
| Declaration (transport by sea) IMDG-Code | No Dangerous Good |
| Declaration (transport by air) IATA-DGR | No Dangerous Good |
| Specifications | |
| Assay (acidimetric) | 97 % |
| Identity (IR) | passes test |
High Purity and Analytical Grade
Acetylenedicarboxylic acid is offered at an exceptional 99% purity, making it ideal for sensitive laboratory and research work. Its analytical grade and consistent composition ensure reliability in organic synthesis and laboratory experiments, supporting precise outcomes in university and specialized research facilities.
Optimal Handling and Storage
This chemical should be handled with care, using appropriate protective clothing, gloves, and eye/face protection. To maintain stability and quality, store the container tightly closed in a dry, well-ventilated area. The product remains stable for up to 24 months when stored under recommended conditions, supporting flexible usage in various laboratory settings.
Versatile Laboratory Applications
Acetylenedicarboxylic acid finds use in research, laboratory procedures, and as a key intermediate in organic synthesis. Its high solubility in water and characteristic of being odorless add to its suitability for controlled experimental environments in academic and industrial research contexts.
FAQ's of Acetylenedicarboxylic acid:
Q: How should Acetylenedicarboxylic acid be safely handled during laboratory use?
A: Always wear suitable protective clothing, gloves, and eye/face protection when handling Acetylenedicarboxylic acid. Avoid direct contact and inhalation of dust. Handle the compound in a well-ventilated area according to established lab safety guidelines.Q: What is the recommended storage process for Acetylenedicarboxylic acid?
A: Keep the product tightly sealed in its original bottle, stored in a dry and well-ventilated place, away from moisture and incompatible substances. Proper storage under these conditions ensures a shelf life of up to 24 months.Q: When is Acetylenedicarboxylic acid typically used in research?
A: This compound is mostly used in university laboratories and research facilities for experiments involving organic synthesis, analytical procedures, and other specialized chemical applications due to its high purity and reliable performance.Q: Where can Acetylenedicarboxylic acid be applied in scientific work?
A: Its application ranges from fundamental research in organic chemistry to laboratory-scale synthesis and academic experiments, making it valuable in research institutions, universities, and professional laboratory settings.Q: What are the benefits of using Acetylenedicarboxylic acid with 99% purity for laboratory applications?
A: High purity ensures minimal interference from impurities, which is essential for achieving accurate, consistent, and reproducible results in quantitative research and analytical experiments.Q: How does the solubility of Acetylenedicarboxylic acid affect its laboratory usage?
A: Being highly soluble in water, it allows for easy preparation of solutions and integration into aqueous chemical reactions, thus broadening its utility in various experimental protocols.Q: What precautions should be taken given its physical and chemical properties?
A: Although it is not classified as hazardous under GHS and is not flammable, appropriate personal protective equipment should be used to prevent contact, and the product should be handled with standard laboratory precautions due to its decomposing nature at elevated temperatures.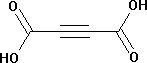
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Laboratory Chemicals Category
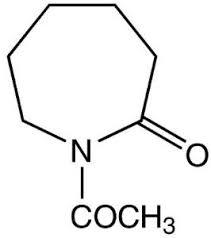
n-ACETYL CAPROLACTAM
Minimum Order Quantity : 25gm Kilograms
Purity(%) : 99%
Grade : Laboratory/Reagent Grade
CAS No : 1888911
Appearance : White to offwhite crystalline powder
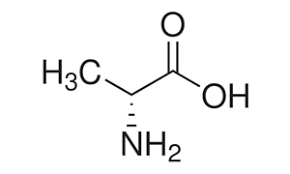
D-ALANINE (for biochemistry)
Price 420 INR
Minimum Order Quantity : 1 Gram
Purity(%) : 99%
Grade : For Biochemistry
CAS No : 338692
Appearance : White crystalline powder
CELLULOSE ACETATE BUTYRATE
Price 3458 INR / Gram
Minimum Order Quantity : 250 Grams
Purity(%) : 99%
Grade : Laboratory Grade
CAS No : 9004368
Appearance : Offwhite Granules or Powder
AMMONIUM PURPURATE solution
Price 703 INR / Milliliter
Minimum Order Quantity : 500 Milliliters
Purity(%) : 98%
Grade : Laboratory Grade
CAS No : 10108642
Appearance : Reddish Violet Solution
We are deals in Lab Chemicals.
"Only deals in retail accepting orders upto 500ml only".
"Only deals in retail accepting orders upto 500ml only".
 |
ALPHA CHEMIKA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese


 Send Inquiry
Send Inquiry