Mercury (II) Nitrate Monohydrate
Mercury (II) Nitrate Monohydrate Specification
- Ph Level
- Acidic in aqueous solution
- Packaging Type
- Sealed bottle, HDPE container, glass bottle
- Density
- 4.3 Gram per cubic centimeter(g/cm3)
- Molecular Weight
- 342.61 g/mol
- Molecular Formula
- Hg(NO3)2H2O
- Purity
- 98%
- Size
- Available in 25g, 100g, 500g, 1kg packs
- Melting Point
- 79C (decomposes)
- Shelf Life
- 2 years when properly stored
- Physical State
- Solid
- Type
- Inorganic Compound
- Application
- Laboratory reagent, chemical synthesis, oxidizing agent
- Appearance
- White to pale yellow crystalline solid
About Mercury (II) Nitrate Monohydrate
This chemical is used in mercuration reactions in chemical laboratory. Holding a highly reputed and trusted market standing, we are engaged in offering our clients Mercury(II) Nitrate Monohydrate. To process this chemical, we use finest quality compounds. It is also used as derivatives for ketone and acetone. Mercury Nitrate Monohydrate is highly demanded in industries like chemical, pharmaceutical and food. Clients can avail this chemical from us in different quantity packaging options. We offer this chemical at affordable prices.
Features:
Longer shelf life
Water soluble
Sharp odor
LC3491 Mercury(II) nitrate monohydrate GR for analysis ACS,Reag. Ph Eur
| Order number | Packaging | Quantity | Price |
| AC30491 | Plastic bottle | 50 g | 22.23 |
| AC30491 | Plastic bottle | 250 g | 63.00 |
| Product information | |
| Grade | ACS,Reag. Ph Eur |
| Hill Formula | HgN2O6 * H2O |
| Chemical formula | HgN2O6 * H2O |
| HS Code | 2852 00 00 |
| EC number | 233-152-3 |
| Molar mass | 342.62 g/mol |
| EC index number | 080-002-00-6 |
| CAS number | 7783-34-8 |
| Chemical and physical data | |
| Solubility | (20 °C) soluble,Hydrolysis |
| Melting point | 79 °C (anhydrous substance) |
| Molar mass | 342.62 g/mol |
| Density | 4.39 g/cm3 (20 °C) |
| Bulk density | 1200 kg/m3 |
| Safety information according to GHS | |
| Hazard Statement(s) | H330: Fatal if inhaled. |
| Precautionary Statement(s) | P280: Wear protective gloves/protective clothing. |
| Signal Word | Danger |
| Hazard Pictogram(s) |
|
| Storage class | Non combustible substances, toxic |
| WGK | WGK 3 highly water endangering |
| Disposal | 20 |
| Safety information | |
| R Phrase | R 26/27/28-33-50/53 |
| S Phrase | S 13-28-45-60-61 |
| Categories of danger | very toxic, dangerous for the environment |
| Hazard Symbol | Very toxic Dangerous for the environment |
| Transport information | |
| Declaration (transport by sea) IMDG-Code | UN 1625 MERCURIC NITRATE, 6.1, II, Marine Pollutant: P, Segregation Group: 7 (Heavy metals and their salts (including their organometallic compounds)), 11 (Mercury and mercury compounds) |
| Declaration (transport by air) IATA-DGR | UN 1625 MERCURIC NITRATE, 6.1, II |
| Toxicological data | |
| LD 50 oral | LD50 rat 26 mg/kg |
| LD 50 dermal | LD50 rat 75 mg/kg |
| Specifications | |
| Assay (complexometric) | 99.0 % |
| Chloride (Cl) | 0.002 % |
| Sulphate (SO4) | 0.002 % |
| Cu (Copper) | 0.0005 % |
| Fe (Iron) | 0.001 % |
| K (Potassium) | 0.005 % |
| Na (Sodium) | 0.005 % |
| Residue after reduction | 0.01 % |
Superior Purity and Versatile Application
Mercury (II) Nitrate Monohydrate is produced to a minimum purity of 98%, making it ideal for analytical and synthetic use. Scientists and researchers value its consistent quality for precise laboratory tasks and advanced chemical synthesis. It also acts as an effective oxidizing agent in specialized reactions, broadening its scope across scientific fields.
Safe Handling and Storage Practices
Due to its high toxicity and chemical reactivity, handling Mercury (II) Nitrate Monohydrate requires strict safety measures. Store the compound in tightly sealed HDPE or glass bottles, away from reducing agents, organic materials, and combustibles. Proper labeling and storage at ambient temperatures extend shelf life and minimize risk.
FAQs of Mercury (II) Nitrate Monohydrate:
Q: How should Mercury (II) Nitrate Monohydrate be handled in the laboratory?
A: Always handle Mercury (II) Nitrate Monohydrate with appropriate personal protective equipment, including gloves and safety goggles. Use in a well-ventilated area or under a chemical fume hood to avoid inhalation, ingestion, or skin contact, as it is highly toxic.Q: What are the typical applications for Mercury (II) Nitrate Monohydrate?
A: This compound is commonly employed as a laboratory reagent, for chemical synthesis, and as an oxidizing agent. Its high purity ensures reliable results in analytical and research settings.Q: When should I avoid using Mercury (II) Nitrate Monohydrate with other chemicals?
A: Avoid combining Mercury (II) Nitrate Monohydrate with strong reducing agents, organic materials, or combustible substances. Such incompatibilities can lead to dangerous reactions, including the release of toxic gases.Q: Where is Mercury (II) Nitrate Monohydrate typically stored and in what packaging?
A: It is best kept in tightly sealed HDPE containers or glass bottles, stored in a cool, dry, and well-ventilated place, away from incompatible materials. Proper storage preserves purity and extends shelf life.Q: What are the hazards and decomposition products of this compound?
A: As a toxic substance, Mercury (II) Nitrate Monohydrate can cause harm if swallowed, inhaled, or absorbed through the skin. Upon decomposition or heating, it can release toxic mercury fumes and nitrogen oxides.Q: How does solubility benefit its use in laboratory processes?
A: Its solubility in water and alcohol makes Mercury (II) Nitrate Monohydrate easy to incorporate into aqueous or organic reactions, facilitating even mixing and efficient chemical reactions.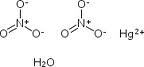

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Mercury Salts Category
Mercuric Oxide Red
Price 100-500 INR / Kilograms
Minimum Order Quantity : 25GM Kilograms
CAS No : 21908532
Purity(%) : 99.72%
Type : Other
Grade : LABORATORY Grade
Mercuric Nitrate
Price 100-500 INR / Kilograms
Minimum Order Quantity : 25 Kilograms
CAS No : 7783348
Purity(%) : 98%
Type : Other
Grade : Laboratory Grade
MERCURIC NITRATE AR (monohydrate)
Price 32504 INR / Gram
Minimum Order Quantity : 500 Grams
CAS No : 7783348
Purity(%) : 99.02%
Type : Other, Analytical Reagent (AR) Monohydrate
Grade : LABORATORY Grade
Ammoniated Mercury
Price 22255 INR / Gram
Minimum Order Quantity : 500 Grams
CAS No : 10124488
Purity(%) : 98%
Type : University Lab Chemicals
Grade : Laboratory Grade
"Only deals in retail accepting orders upto 500ml only".
 |
ALPHA CHEMIKA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese


 Send Inquiry
Send Inquiry






