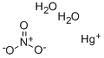
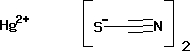Mercuric Nitrate
Price 100-500 INR/ Kilograms
Mercuric Nitrate Specification
- Packaging Type
- Bottle
- Density
- 4.3 Gram per cubic centimeter(g/cm3)
- Molecular Weight
- 324.6 g/mol
- Molecular Formula
- Hg(NO3)2
- Physical State
- Solid
- Storage Instructions
- Store in a cool, dry, well-ventilated place. Keep container tightly closed and away from incompatible materials.
- Purity
- 98%
- Melting Point
- 70 C decomposes
- Shelf Life
- 1 year (under recommended storage conditions)
- Boiling point
- Decomposes before boiling
- Size
- As required/by demand
- Ph Level
- Acidic
- Usage
- Used in chemical analysis, as a laboratory reagent, and for the preparation of other mercury compounds.
- CAS No
- 7783-34-8
- Grade
- Laboratory Grade
- Type
- Other
- Application
- Laboratory Reagent, Analytical Chemistry, Preparation of other Mercury Compounds
- Appearance
- White Crystalline Powder
- Purity(%)
- 98%
- UN Number
- UN1625
- Odor
- Odorless
- Solubility
- Soluble in water and dilute acids
- HS Code
- 28439000
- Reactivity
- Reacts with organic materials and reducing agents
- Hazard Statement
- Toxic by inhalation, ingestion, and in contact with skin. Strong oxidizer.
- Decomposition Products
- Releases toxic nitrogen oxides and mercury vapors on decomposition
Mercuric Nitrate Trade Information
- Minimum Order Quantity
- 25 Kilograms
- FOB Port
- Nhva sheva
- Payment Terms
- Cash Advance (CA), Cash in Advance (CID), Cheque
- Supply Ability
- 1000 Kilograms Per Day
- Delivery Time
- 1 Week
- Sample Available
- Yes
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Packaging Details
- 500GM, 1KG, 2.5KG, 5KG, 25KG, 50 KG export worthy Packing
- Main Domestic Market
- All India
- Certifications
- ISO 9001:2008, GMP
About Mercuric Nitrate
Specifications of Mercuric Nitrate Extra Pure
- Chemical Names : Mercury Nitrate, Mercury(II) Nitrate, Mercury Dinitrate, Citrine Ointment,
- PubChem CID : 24864
- EC Number : 233-152-3
- UN/NA Number : 1625
- Molecular Formula : HgN2O6
- Molecular Weight : 342.62 g/mol
- Appearance : White crystalline soild
- Minimum Assay : 98%
- Physical State : Powder
- Usage : Industrial, Laboratory
- Solubility : Soluble in water & nitric acid & insoluble in alcohol
- Grade Standard : Reagent grade, Analytical grade, Technical grade
- M.P. : 79° C
- Density : 4.3 @20° C
- Un No. 6.1/1625
- TARIFF : 2834 29 90
- R : 26/27/28-33 S : 13-28.1-45 F : 20
Maximum Limits of Impurities 10 x
- Chloride(CI) 0.005%
- Sulphate(SO4) 0.01%
- Iron(Fe) 0.005%
- Mercury(I)(Hg) 0.5%
- Potassium(K) 0.01%
- Sodium(Na) 0.01%
Key Features and Specifications
Mercuric nitrate is supplied as a white, odorless crystalline powder with a density of 4.3 g/cm3 and an acidic pH. With a melting point of 70C (decomposing upon melting), it decomposes before boiling. The product is available in various bottle sizes as required, with a shelf life of one year under recommended storage. It is soluble in water and dilute acids, making it suitable for a wide range of laboratory applications.
Safe Handling and Storage Guidelines
Due to its hazardous properties-including toxicity and strong oxidizing behavior-mercuric nitrate should be handled with personal protective equipment in a well-ventilated laboratory. Store it tightly sealed in a cool, dry location, away from incompatible substances such as organic materials and reducing agents. Always follow best industrial hygiene practices when using or storing this chemical.
Applications and Professional Uses
Mercuric nitrate is utilized primarily in chemical analysis, as a laboratory reagent, and in the synthesis of other mercury-based compounds. Its reliability and high purity make it indispensable in analytical chemistry, and its reactivity aids in various specialized laboratory processes. Proper protocols should be followed to maximize safety and benefit from its effectiveness.
FAQ's of Mercuric Nitrate:
Q: How should mercuric nitrate be safely stored?
A: Mercuric nitrate should be kept in a tightly closed container in a cool, dry, and well-ventilated place. It must be stored away from organic materials and reducing agents to avoid hazardous reactions.Q: What is the recommended process for using mercuric nitrate in the laboratory?
A: Use appropriate personal protective equipment such as gloves, lab coat, and goggles. Work in a well-ventilated area, avoid direct contact, and follow all institutional protocols for handling toxic and oxidizing substances.Q: When does mercuric nitrate decompose, and what are its decomposition products?
A: Mercuric nitrate decomposes at around 70C and does not boil but breaks down before reaching a boiling point. The decomposition releases toxic nitrogen oxides and mercury vapors.Q: Where is mercuric nitrate typically applied in professional settings?
A: It is mainly used in analytical chemistry, as a laboratory reagent, and for the preparation of other mercury compounds by professionals in research, industrial, and educational laboratories.Q: What are the hazards associated with mercuric nitrate and how can risks be minimized?
A: Mercuric nitrate is toxic by inhalation, ingestion, and skin contact, and is a strong oxidizer. Minimize risk by using the chemical in a fume hood, wearing suitable protective gear, and storing it properly.Q: Can mercuric nitrate be used for the preparation of other mercury compounds?
A: Yes, due to its high reactivity and purity, mercuric nitrate is widely used as a precursor in synthesizing a variety of other mercury compounds in laboratory settings.Q: What benefits does mercuric nitrate offer for analytical chemistry?
A: With high purity, solubility, and predictable reactivity, mercuric nitrate allows for precise analyses and preparation of standard solutions, making it a dependable reagent for various chemical tests.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Mercury Salts Category
MERCURIC IODIDE RED AR
Price 14999.00 INR / Gram
Minimum Order Quantity : 10 Grams
CAS No : 7774290
Grade : LABORATORY Grade
Type : Other, Analytical Reagent (AR)
Appearance : Reddish orange powder
Ammoniated Mercury
Price 22255 INR / Gram
Minimum Order Quantity : 500 Grams
CAS No : 10124488
Grade : Laboratory Grade
Type : University Lab Chemicals
Appearance : White or yellowish white, odorless, amorphous powder
Mercury (I) Nitrate Dihydrate
Price 35994 INR / Gram
Minimum Order Quantity : 500 Grams
CAS No : 14836603
Grade : Laboratory Grade
Type : University Lab Chemicals
Appearance : White to pale yellow crystalline solid
Mercury (II) Thiocyanate
Price 35603 INR / Gram
Minimum Order Quantity : 500 Grams
CAS No : 592858
Grade : Laboratory Grade
Type : University Lab Chemicals
Appearance : White to grey crystalline powder
"Only deals in retail accepting orders upto 500ml only".
 |
ALPHA CHEMIKA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese


 Send Inquiry
Send Inquiry