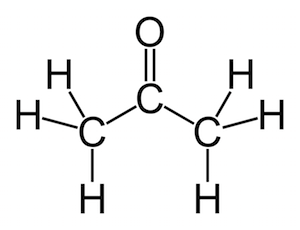
Di-potassium Oxalate Monohydrate
Price 835 INR/ Gram
Di-potassium Oxalate Monohydrate Specification
- Purity
- 99%
- Melting Point
- Not applicable (decomposes)
- Molecular Weight
- 228.18 g/mol
- Physical State
- Solid
- Molecular Formula
- K2C2O4H2O
- CAS No
- 6487-48-5
- Grade
- Laboratory Grade
- Type
- Inorganic Salt
- Application
- Analytical Reagent, Laboratory Use, Chemical Synthesis
- Appearance
- White Crystalline Powder
- Purity(%)
- 99%
- Shelf Life
- 5 Years
- Size
- Customizable as per requirement
- Solubility
- Soluble in water
- Odor
- Odorless
- Usage
- Laboratory reagent, Metal Treatment, Textile Industry
- Storage Instructions
- Store in a cool, dry place, keep container tightly closed
- HS Code
- 29171110
- EC No.
- 208-506-9
- Packaging Type
- HDPE Bottle, Drum, Customized Packaging
- Ph Level
- 7.0 (1% solution at 20C)
- pH of 5% Solution
- 6.5 - 7.5
- Moisture Content
- 1%
- Density
- 2.0 g/cm
Di-potassium Oxalate Monohydrate Trade Information
- Minimum Order Quantity
- 500 Grams
- Supply Ability
- 50000 Grams Per Week
- Delivery Time
- 4-5 Days
About Di-potassium Oxalate Monohydrate
Di-potassium Oxalate Monohydrate
LC5107 di-Potassium oxalate monohydrate
or analysis EMSURE
Order number Packaging Quantity Price
AC32107 Plastic bottle 250 g 18.225
AC32107 Plastic bottle 1 kg
54.00
Product information
Synonyms Oxalic acid dipotassium salt monohydrateC2K2O4 * H2O
Hill Formula K2C2O4 * H2O
Chemical formula K2C2O4 * H2O
HS Code 2917 11 00
EC number 209-506-8
Molar mass 184.24 g/mol
EC index number 184.24 g/mol
CAS number 6487-48-5
Chemical and physical data
Solubility 360 g/l (20 C) (anhydrous substance)
Molar mass 184.24 g/mol
Density 2.13 g/cm3 (20 C)
Bulk density 700 - 1100 kg/m3
pH value 7.0 - 8.5 (50 g/l, H2O, 20 C)
Vapor pressure (20 C)
Safety information according to GHS
Hazard Statement(s) H312: Harmful in contact with skin.
H302: Harmful if swallowed.
Precautionary Statement(s) P302 + P352: IF ON SKIN: Wash with plenty of soap and water.
Signal Word Warning
Hazard Pictogram(s)
Storage class 10 - 13 Other liquids and solids
WGK WGK 1 slightly water endangering
Disposal 3
Relatively unreactive organic reagents should be collected in container A. If halogenated, they should be collected in container B. For solid residues use container C.
Safety information
R Phrase R 21/22
Harmful in contact with skin and if swallowed.
S Phrase S 24/25
Avoid contact with skin and eyes.
Categories of danger harmful
Hazard Symbol Harmful
Toxicological data
LD 50 oral LD50 rat 660 mg/kg
Specifications
Assay (oxidimetric) 99.5 - 101.0 %
Insoluble matter 0.01 %
Neutrality passes test
Chloride (Cl) 0.001 %
Sulphate (SO4) 0.01 %
Heavy metals (as Pb) 0.001 %
NH4 (Ammonium) 0.002 %
Fe (Iron) 0.0005 %
Na (Sodium) 0.02 %
Substances darkened by hot sulfric acid passes test
Versatile Laboratory and Industrial Applications
Di-potassium Oxalate Monohydrate serves as an essential analytical reagent across various sectors. Its high purity and consistent solubility make it ideal for laboratory experiments, chemical synthesis, and precise metal treatment. Textile processors also value its effective functional properties and reliable performance in diverse processes.
Optimal Storage and Longevity
With a shelf life of five years, this compound retains its quality when stored in cool, dry conditions and in tightly closed containers. Its moisture content remains 1%, ensuring the product's stability and usability even with long-term storage in industrial or research settings.
Customizable Packaging Solutions
This product is offered in an array of packaging options-including HDPE bottles, drums, and customized containers-to accommodate varying volume and operational requirements. Clients can select the size and container type most compatible with their workflow and storage capacity.
FAQ's of Di-potassium Oxalate Monohydrate:
Q: How should Di-potassium Oxalate Monohydrate be stored for maximum shelf life?
A: To preserve its quality, Di-potassium Oxalate Monohydrate should be kept in a cool, dry place. Ensure the container remains tightly closed to prevent moisture absorption. Properly stored, its shelf life can extend up to five years.Q: What are the primary uses of Di-potassium Oxalate Monohydrate in industries?
A: This compound is widely used as a laboratory reagent, in metal treatment processes, the textile industry, and for chemical synthesis. Its high purity makes it suitable for precise analytical applications and research settings.Q: When is customized packaging recommended for this product?
A: Customized packaging is ideal when specific quantities or unique container types are required for your workflow or storage constraints. The product can be supplied in HDPE bottles, drums, or tailored options based on customer needs.Q: Where is Di-potassium Oxalate Monohydrate most commonly applied?
A: It is commonly found in laboratories for analytical work, industrial facilities conducting metal treatment, and textile manufacturing plants benefiting from its chemical properties and ease of solubility.Q: What is the benefit of using this product as an analytical reagent?
A: Its 99% purity and stable pH profile make it highly reliable for analytical tasks, resulting in consistent, reproducible results in research and quality control processes.Q: How does solubility in water affect its process use?
A: Its excellent water solubility allows it to be easily prepared into solutions of precise concentration, facilitating accurate experimentation and industrial processing.Q: What precautions should be taken during handling?
A: Handle with care in a well-ventilated area, and always use appropriate personal protective equipment. Avoid ingestion or inhalation of dust, and keep away from incompatible substances.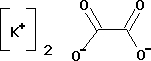

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Laboratory Chemicals Category
ACETONE ALCOHOL
Price 355 INR / Milliliter
Minimum Order Quantity : 500 Milliliters
Type : University Lab Chemicals
Grade : LR
Purity(%) : 99%
Appearance : Clear colorless liquid
DIGITONIN 99% AR
Price 161236 INR / Gram
Minimum Order Quantity : 5 Grams
Type : University Lab Chemicals
Grade : AR
Purity(%) : 99%
Appearance : White Powder
D-CAMPHOR - 10 - SULPHONIC ACID
Type : Other, Chemical Compound
Grade : Industrial
Purity(%) : 98%
Appearance : White Crystalline Powder
Hydroxyphenylacetonitrile Chemical
Type : Industrial Lab Chemicals
Grade : Industrial
Purity(%) : 99.99
"Only deals in retail accepting orders upto 500ml only".
 |
ALPHA CHEMIKA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese


 Send Inquiry
Send Inquiry