Phenylpropionitrile Chemical
MOQ : 500 Milliliters
Phenylpropionitrile Chemical Specification
- Flash Point
- 104C
- Molecular Weight
- 131.18 g/mol
- Size
- Custom packaging available
- Melting Point
- -30C
- Boiling point
- 232C
- Packaging Type
- Bottle, Drum
- CAS No
- 645-59-0
- Shelf Life
- 24 Months
- Density
- 0.995 Gram per cubic centimeter(g/cm3)
- Molecular Formula
- C9H9N
- Usage
- Used as intermediate in organic synthesis and drug manufacturing
- Purity
- 98%
- Storage Instructions
- Store in a cool, dry, and well-ventilated place. Keep container tightly closed.
- Physical State
- Liquid
- Grade
- Laboratory Grade
- Type
- Chemical Compound
- Application
- Organic Synthesis, Pharmaceutical Intermediate
- Appearance
- Colorless to pale yellow liquid
- Purity(%)
- 98%
- Odor
- Aromatic odor
- EC Number
- 211-448-7
- Refractive Index
- 1.527 - 1.529 at 20°C
- Hazard Class
- Harmful if inhaled, irritant
- Assay
- >=98%
- Solubility
- Insoluble in water, soluble in organic solvents
- HS Code
- 29269000
- Compatibility
- Compatible with most organic solvents
- Stability
- Stable under recommended storage conditions
Phenylpropionitrile Chemical Trade Information
- Minimum Order Quantity
- 500 Milliliters
- Supply Ability
- 50000 Milliliters Per Week
- Delivery Time
- 4-5 Days
About Phenylpropionitrile Chemical
LC4665 3-Phenylpropionitrile
for synthesis
Order number Packaging Quantity Price
AC31665 Glass bottle 10 ml 43.43
Product information
Synonyms Hydrocinnamonitrile, Benzylacetonitrile
Hill Formula C9H9N
HS Code 2926 90 95
EC number 211-447-8
Molar mass 131.17 g/mol
CAS number 645-59-0
Chemical and physical data
Solubility 1.68 g/l (25 C) (calculated)
Melting point -2 C
Molar mass 131.17 g/mol
Density 1.001 g/cm3 (20 C)
Boiling point 114 - 116 C (13.3 hPa)
Flash point 110 C
Safety information
R Phrase R 25
Toxic if swallowed.
S Phrase S 37-45
Wear suitable gloves.In case of accident or if you feel unwell, seek medical advice immediately (show the label where possible).
Categories of danger toxic
Hazard Symbol
Toxic
Storage class Combustible substances, toxic
WGK WGK 3 highly water endangering
Disposal 9
Carcinogenic compounds and flammable compounds labelled "Highly toxic" or "Toxic": container F. Alkyl sulfates are carcinogenic; Take particular care to avoid inhalation and skin contact. To neutralize alkyl sulfates, add dropwise (from a dropping funnel) to concentrated ice-cool Ammonia solution (Cat.No. 105426) with vigorous stirring. Before placing in container D, check the pH with pH Universal indicator strips (Cat.No. 109535).
Transport information
Declaration (transport by sea) IMDG-Code UN 3276 NITRILES, TOXIC, LIQUID, N.O.S.(3-PHENYLPROPIONITRILE), 6.1, II
Declaration (transport by air) IATA-DGR UN 3276 NITRILES, TOXIC, LIQUID, N.O.S.(3-PHENYLPROPIONITRILE), 6.1, II
Specifications
Assay (GC, area%) 99 %
Identity (IR) passes test
Versatile Applications in Synthesis and Pharmaceuticals
Phenylpropionitrile finds significant use as an intermediate in organic synthesis and the pharmaceutical industry. Its chemical stability and high purity make it ideal for processes requiring reliable reactants. The compound facilitates the manufacturing of advanced intermediates and active pharmaceutical ingredients, supporting research and production efficiency.
Safe Storage and Handling Recommendations
To maintain its stability and quality, phenylpropionitrile should be stored in a cool, dry, well-ventilated area, with the container tightly closed. Avoid exposure to heat and ignition sources, as the liquid has a flash point of 104C. Following appropriate handling protocols minimizes inhalation risks, ensuring safe usage in laboratory and industrial environments.
FAQ's of Phenylpropionitrile Chemical:
Q: How should phenylpropionitrile be stored to ensure maximum shelf life?
A: Store phenylpropionitrile in a cool, dry, and well-ventilated place, keeping the container tightly closed. These conditions help maintain its stability and extend its shelf life to 24 months.Q: What are the main applications of phenylpropionitrile in industry?
A: This compound is primarily used as an intermediate in organic synthesis and pharmaceutical manufacturing, supporting the production of various complex chemicals and active drug ingredients.Q: Where can phenylpropionitrile be sourced in India?
A: Phenylpropionitrile is available through several channels in India, including dealers, exporters, manufacturers, retailers, suppliers, and traders. Packaging is usually offered in bottles or drums with custom sizes available.Q: What safety measures should be followed when handling phenylpropionitrile?
A: Since phenylpropionitrile is harmful if inhaled and can cause irritation, use it in a well-ventilated area, wear appropriate personal protective equipment, and avoid direct exposure. Ensure containers remain tightly closed when not in use.Q: How is phenylpropionitrile typically used in organic synthesis?
A: Phenylpropionitrile acts as a valuable intermediate for introducing a nitrile functional group into target compounds, facilitating various transformations crucial in the synthesis of pharmaceuticals and specialty chemicals.Q: What benefits does phenylpropionitrile offer in pharmaceutical manufacturing?
A: The compound's high purity (98%) and compatibility with organic solvents make it suitable for precise synthetic steps in drug development, ensuring consistent quality and efficiency in production processes.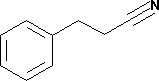
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Laboratory Chemicals Category
N N-DIETHYLANILINE (for synthesis)
Price 916 INR / Milliliter
Minimum Order Quantity : 500 Milliliters
Purity(%) : 99 %
Grade : AR
Type : University Lab Chemicals
CAS No : 91667
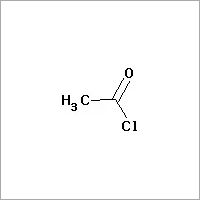
Acetyl Chloride
Price 1025 INR
Minimum Order Quantity : 500 Milliliters
Purity(%) : 99%
Grade : Industrial Grade
Type : Industrial Lab Chemicals
CAS No : 75365
CALCIUM FLUORIDE (practical)
Price 1051 INR / Gram
Minimum Order Quantity : 500 Grams
Purity(%) : 95%
Grade : Laboratory Grade
Type : Other, Inorganic Compound
CAS No : 7789755
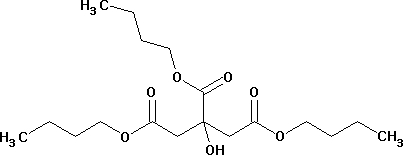
Tributyl citrate
Minimum Order Quantity : 500 Milliliters
Purity(%) : 99.99
Grade : Industrial
Type : Industrial Lab Chemicals
We are deals in Lab Chemicals.
"Only deals in retail accepting orders upto 500ml only".
"Only deals in retail accepting orders upto 500ml only".
 |
ALPHA CHEMIKA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese


 Send Inquiry
Send Inquiry