CADMIUM ACETATE
Price 1902 INR/ Gram
MOQ : 500 Grams, ,
CADMIUM ACETATE Specification
- Molecular Weight
- 266.53 g/mol
- Density
- 2.34 Gram per cubic centimeter(g/cm3)
- Physical State
- Powder
- Boiling point
- Decomposes
- Packaging Type
- Bottle
- Size
- 100 g, 500 g, 1 kg
- Usage
- Laboratory, Research, Industrial
- Shelf Life
- 24 Months
- Melting Point
- 240 C
- Molecular Formula
- C4H6CdO4
- Purity
- 99%
- Storage Instructions
- Store in a cool, dry place, tightly closed
- CAS No
- 5743-04-4
- Grade
- Analytical Reagent
- Type
- Industrial Lab Chemicals
- Application
- Used In Laboratory
- Appearance
- White Crystalline Powder
- Purity(%)
- 99%
- Moisture Content
- <0.5%
- Odor
- Odorless
- Hazard Statement
- Toxic if swallowed, may cause cancer
- HS Code
- 29152990
- Solubility
- Soluble in water and alcohol
- Decomposition Temperature
- >240 °C
- Color
- White
- Reactivity
- Stable under recommended storage conditions
- Assay
- >99%
- Grade Standard
- Technical Grade, Laboratory Grade
CADMIUM ACETATE Trade Information
- Minimum Order Quantity
- 500 Grams, ,
- Supply Ability
- 50000 Grams, , Per Week
- Delivery Time
- 4-5 Days
About CADMIUM ACETATE
CADMIUM ACETATE
- CADMIUM ACETATE EXTRA PURE 100 gm
- (CH3COO)2Cd.2H2O M.W. 266.52 250 gm
- Minimum assay 500 gm
- (complexometric) 98% 10 X 500 gm
- Maximum limits of Impurities :
- Chloride(Cl) 0.005%
- Nitrate(NO3) 0.005%
- Sulphate(SO4) 0.01%
- Iron(Fe) 0.002%
- Potassium(K) 0.01%
- Sodium(Na) 0.01%
- Zinc(Zn) 0.02%
- CAS No. 5743-04-4 Un No. 6.1/2570 TARIFF : 2915 29 90
- R : 20/21/22 S : 22 F : 15
High-Purity Cadmium Acetate for Precise Applications
Cadmium Acetate is manufactured to strict analytical reagent standards with an assay greater than 99%. Its stable composition makes it ideal for a range of laboratory and industrial experiments. The product's white, crystalline appearance, high solubility in water and alcohol, and virtually moisture-free state ensure reliable results in sensitive procedures.
Safe Handling and Storage Recommendations
Since Cadmium Acetate is toxic if swallowed and may pose long-term health hazards, proper storage is paramount. Keep the compound in a tightly closed bottle in a cool, dry place away from incompatible substances. Always use personal protective equipment when handling this product to ensure maximum safety and preservation of the chemical's properties.
FAQ's of CADMIUM ACETATE:
Q: How should Cadmium Acetate be stored to maintain its stability and purity?
A: Cadmium Acetate should be stored in a tightly closed bottle, placed in a cool and dry environment. This preserves its analytical reagent grade quality and ensures a long shelf life of up to 24 months. Proper storage also prevents degradation and moisture uptake.Q: What is the recommended process for dissolving Cadmium Acetate in laboratory solutions?
A: To prepare solutions, add the required amount of Cadmium Acetate to either water or alcohol while stirring. The compound is readily soluble in these solvents, which makes solution preparation straightforward and efficient.Q: Where is Cadmium Acetate commonly used?
A: Cadmium Acetate finds applications in laboratories, research institutions, and various industrial processes. It is supplied across India by manufacturers, exporters, and dealers for analytical, research, and technical uses.Q: When is Cadmium Acetate considered necessary in laboratory procedures?
A: Cadmium Acetate is selected when high-purity cadmium sources are required for analytical testing, synthesis, or when a consistent, stable reactant is needed in laboratory or industrial research.Q: What are the safety considerations and hazard statements associated with Cadmium Acetate?
A: Cadmium Acetate is toxic if swallowed and may cause cancer with prolonged exposure. Always handle it with appropriate protective gear, avoid ingestion and inhalation, and work in well-ventilated areas or fume hoods.Q: How does Cadmium Acetate benefit research and analytical processes?
A: Its high purity and stable crystalline form ensure accurate and repeatable results in quantitative analysis and specialized synthesis, making it a valuable reagent for research and lab work.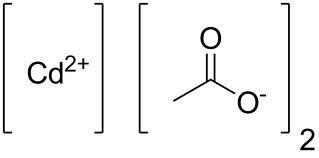
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Laboratory Chemicals Category
2- AMINOPYRAZINE
Price 16138 INR / Gram
Minimum Order Quantity : 25 Grams
Grade : Laboratory Grade
Purity(%) : 97%
Type : Other, Chemical Compound
Appearance : White to offwhite crystalline solid
L-Cysteine hydrochloride monohydrate
Price 3578 INR / Gram
Minimum Order Quantity : 500 Grams
Grade : Laboratory Grade
Purity(%) : 98%
Type : Other, Amino Acid
Appearance : White crystalline powder
Pyruvaldehyde 1,1-dimethyl acetal
Minimum Order Quantity : 500 Milliliters
Grade : Industrial
Purity(%) : 99.99
Type : Industrial Lab Chemicals
ANILINE HYDROCHLORIDE
Grade : Industrial
Purity(%) : 99%
Type : Chemical Compound, Other
Appearance : White crystalline powder
We are deals in Lab Chemicals.
"Only deals in retail accepting orders upto 500ml only".
"Only deals in retail accepting orders upto 500ml only".
 |
ALPHA CHEMIKA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese


 Send Inquiry
Send Inquiry