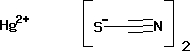Mercurous Nitrate
Price 100-500 INR/ Kilograms
Mercurous Nitrate Specification
- Packaging Type
- Bottle
- Storage Instructions
- Store in cool, dry place, away from light
- Physical State
- Powder
- Ph Level
- Slightly acidic in solution
- Molecular Formula
- Hg2(NO3)2
- Size
- Custom packaging available
- Purity
- 95%
- Density
- 5.22 Gram per cubic centimeter(g/cm3)
- Melting Point
- 70 C (decomposition)
- Usage
- For laboratory and research use only
- Molecular Weight
- 342.61 g/mol
- Shelf Life
- 24 months (when properly stored)
- CAS No
- 14836 - 60 - 3
- Grade
- Laboratory Chemicals
- Type
- Other
- Application
- Analytical reagent, preparation of other mercury compounds
- Appearance
- Colourless to white crystalline powder
- Purity(%)
- 95%
- Transport Information
- Handle as toxic chemical, follow hazardous material protocols
- Reactivity
- Reacts with acids and oxidizing agents
- Hazards
- Highly toxic, avoid contact
- Odor
- Odorless
- Regulatory Status
- Requires special permit for handling
- UN Number
- UN 1645
- HS Code
- 28439010
- Solubility
- Soluble in water, decomposes
- Stability
- Decomposes gradually on exposure to air
Mercurous Nitrate Trade Information
- Minimum Order Quantity
- 25 gm Kilograms
- FOB Port
- Nhav Sheva
- Payment Terms
- Cash in Advance (CID), Cheque, Cash Advance (CA)
- Supply Ability
- 1000 Kilograms Per Day
- Delivery Time
- 1 Week
- Sample Available
- Yes
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Packaging Details
- 25 gm , 100 gm , 500 gm , 25kg
- Main Domestic Market
- All India
- Certifications
- Specifications :- Synonyms : Mercurous Nitrate Dihydrate, Mercurous Nitrate CBNumber : CB7447657 Molecular Formula : HgNO3 Formal Weight : 262.59 Appearance : Colourless to white crystalline powder Odor : slight odor of Nitric Acid
About Mercurous Nitrate
It is used in the preparation of other mercury compounds. We have established ourselves as a quality focused organization engaged in offering Mercurous Nitrate. It is used in mercuration reaction for chemistry and scientific laboratory. We use premium quality compounds, sourced from trusted vendors to process this chemical. Mercurous Nitrate is widely used in chemical, pharmaceutical and bio chemistry. Clients can avail our chemical in different quantity packaging options.
Features:
- Colorless white crystals
- Longer shelf life
- Soluble in water
Specifications :-
- Synonyms : Mercurous Nitrate Dihydrate, Mercurous Nitrate
- CBNumber : CB7447657
- Molecular Formula : HgNO3
- Formal Weight : 262.59
- Appearance : Colourless to white crystalline powder
- Odor : slight odor of Nitric Acid
- Melting Point : 70C
- Density : 4.78 g/ml
- Purity : 95%
Features :-
- Colorless white crystals
- Longer shelf life
- Soluble in water
Key Properties and Applications
Mercurous Nitrate stands out for its use as an analytical reagent and for synthesizing various mercury-based compounds. Offered in laboratory grade and at 95% purity, it is commonly provided as a colorless to white crystalline powder. Its slightly acidic aqueous solution and specific reactivity profile make it suitable for specialized research and laboratory tasks.
Handling and Safety Information
Due to its high toxicity and gradual decomposition upon air exposure, Mercurous Nitrate requires meticulous handling. Proper personal protective equipment (PPE), adherence to hazardous material protocols, and secure storage are essential. Always store the material in cool, dry conditions, away from light, and ensure all applicable regulations and special permits are in place before use.
FAQ's of Mercurous Nitrate:
Q: How should Mercurous Nitrate be safely handled in the laboratory?
A: Due to its highly toxic nature, Mercurous Nitrate should be handled with proper personal protective equipment, such as gloves and eye protection. Work within a well-ventilated fume hood, avoid any skin or eye contact, and strictly follow hazardous material protocols. Only trained personnel with the required permits should handle this chemical.Q: What are the main uses of Mercurous Nitrate?
A: Mercurous Nitrate is primarily used as an analytical reagent in laboratory settings and for preparing other mercury-based compounds. It is intended strictly for laboratory and research purposes and should not be used for any other applications.Q: When does Mercurous Nitrate begin to decompose, and what precautions should be taken?
A: Mercurous Nitrate decomposes gradually when exposed to air and also decomposes upon melting at 70C. To minimize decomposition, always store it in a tightly sealed bottle in a cool, dry place, away from light, and use it promptly after opening.Q: Where should Mercurous Nitrate be stored to maintain its stability?
A: Store Mercurous Nitrate in a cool, dry place, away from direct light and incompatible substances such as acids and oxidizing agents. Use airtight, chemical-resistant bottles and ensure the storage area is designated for hazardous materials.Q: What is the process for acquiring and legally transporting Mercurous Nitrate?
A: Mercurous Nitrate is regulated as a hazardous material and requires a special permit for purchase and transportation. It must be packaged according to hazardous material guidelines, display UN Number 1645, and should always be handled by authorized and trained personnel following proper documentation protocols.Q: How does the solubility of Mercurous Nitrate influence its laboratory applications?
A: Mercurous Nitrate is soluble in water but decomposes as it dissolves, producing a slightly acidic solution. This property is considered during analytical processes, and immediate use after preparation is recommended to retain accuracy in results.Q: What is the benefit of using laboratory-grade Mercurous Nitrate with 95% purity?
A: The high purity level ensures consistent and reliable outcomes in analytical procedures and compound synthesis. Laboratory grade Mercurous Nitrate minimizes contamination and enhances the accuracy of experimental results, making it ideal for research and specialized laboratory work.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Mercury Salts Category
Mercuric Oxide Red
Price 100-500 INR / Kilograms
Minimum Order Quantity : 25GM Kilograms
Purity(%) : 99.72%
CAS No : 21908532
Type : Other
Grade : LABORATORY Grade
Mercurous Chloride
Price 100-500 INR / Kilograms
Minimum Order Quantity : 25GM Kilograms
Purity(%) : 99%
CAS No : 10112911
Type : Other
Grade : Laboratory Grade
Mercuric Sulphate
Price 100-500 INR / Kilograms
Minimum Order Quantity : 25GM Kilograms
Purity(%) : 98%
CAS No : 7783359
Type : Other, Inorganic Chemical Compound
Grade : Laboratory Grade
Mercury (II) Thiocyanate
Price 35603 INR / Gram
Minimum Order Quantity : 500 Grams
Purity(%) : 97%
CAS No : 592858
Type : University Lab Chemicals
Grade : Laboratory Grade
"Only deals in retail accepting orders upto 500ml only".
 |
ALPHA CHEMIKA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese


 Send Inquiry
Send Inquiry