Tetra-n-butylammonium borohydride
Tetra-n-butylammonium borohydride Specification
- Storage Instructions
- Dry Place
- Usage
- Lab Chemicals
- Grade
- Industrial
- Type
- Industrial Lab Chemicals
- Application
- Lab Chemicals
- Purity(%)
- 99.99
About Tetra-n-butylammonium borohydride
LC5198 Tetra-n-butylammonium borohydridefor synthesis Order number Packaging Quantity Price AC32198 Glass bottle 10 g 53.10 Product information Synonyms Tetra-n-butylammonium-tetrahydridoborate Hill Formula C16H40BN HS Code 2942 00 00 EC number 251-658-2 Molar mass 257.31g/mol CAS number 33725-74-5 Chemical and physical data Melting point 124-129C Molar mass 257.31g/mol Flash point 140C Safety information according to GHS Hazard Statement(s) H261: In contact with water releases flammable gases.H314: Causes severe skin burns and eye damage. Precautionary Statement(s) P280: Wear protective gloves/ protective clothing/ eye protection/ face protection.P301 + P330 + P331: IF SWALLOWED: rinse mouth. Do NOT induce vomiting.P305 + P351 + P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing.P402 + P404: Store in a dry place. Store in a closed container. Signal Word Danger Hazard Pictogram(s) Storage class 4.3 Materials that form flammble gases in contact with water WGK WGK 3 highly water endangering Disposal 26Alkali and alkaline-earth metals should be taken up in an inert solvent and neutralized by adding 2-propanol (Cat. No. 100995) dropwise with stirring. Should the reaction be more violent than expected, conversion should be carried out with tert-butanol or octanol. Caution: This reaction produces hydrogen, which can form explosive mixtures; take necessary precautions. When the reaction has ceased, add water dropwise; neutralize; container D. In the case of alkali or alkaline-earth borohydrides, add methanol (Cat. No. 106008) with stirring; in the case of alkali or alkaline-earth amides and hydrides, organoaluminium and organotin hydrides, add 2-propanol (Cat. No. 100995) dropwise. The substances, which are usually in solid form, should be previously suspended in an ether! When the respective reaction has ceased, hydrolize with water; then neutralize. Container D or E. Lithium aluminium hydride must also be destroyed by slurrying in an ether. Under an inert gas and with thorough stirring, add dropwise a 1: 4 mixture of ethyl acetate (Cat. No. 822277) and the ether used to prepare the slurry. Always ensure thorough mixing (stirring)! Container A. Safety information R Phrase R 34Causes burns. S Phrase S 6-26-36/37/39-45Keep under protective gas.In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.Wear suitable protective clothing, gloves and eye/face protection.In case of accident or if you feel unwell, seek medical advice immediately (show the label where possible). Categories of danger corrosive Hazard Symbol Corrosive Transport information Declaration (transport by sea) IMDG-Code UN 3131 WATER-REACTIVE SOLID, CORROSIVE, N.O.S.(TETRA-N-BUTYLAMMONIUMBOROHYDRIDE), 4.3 (8), II, Segregation Group: 18 (Alkalis) Declaration (transport by air) IATA-DGR UN 3131 WATER-REACTIVE SOLID, CORROSIVE, N.O.S.(TETRA-N-BUTYLAMMONIUMBOROHYDRIDE), 4.3 (8), II Specifications Assay(hypochlorite) 97% Melting range - lower value 124C - upper value 129C Identity(IR) passes testFAQs of Tetra-n-butylammonium borohydride:
Q: What is the purity percentage of Tetra-n-butylammonium borohydride?
A: The purity percentage of Tetra-n-butylammonium borohydride is 99.99%.Q: What type of chemical is Tetra-n-butylammonium borohydride?
A: Tetra-n-butylammonium borohydride is classified as an Industrial Lab Chemical.Q: What is the recommended storage condition for Tetra-n-butylammonium borohydride?
A: It is recommended to store Tetra-n-butylammonium borohydride in a dry place.Q: In what grade is Tetra-n-butylammonium borohydride available?
A: Tetra-n-butylammonium borohydride is available in Industrial grade.Q: What is the primary application of Tetra-n-butylammonium borohydride?
A: The primary application of Tetra-n-butylammonium borohydride is as a lab chemical.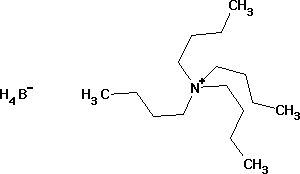

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Laboratory Chemicals Category
p-AMINO ANTIPYRINE
Price 1145 INR
Minimum Order Quantity : 25 Grams
Purity(%) : 99%
Type : Industrial Lab Chemicals
Grade : Laboratory Grade
Application : Laboratory, Analytical Reagent, Coloring Agent in Biochemical Applications
P - BROMO ACETANILIDE
Price 6463 INR / Gram
Minimum Order Quantity : 500 Grams, ,
Purity(%) : 98%
Type : Other, Organic compound
Grade : Laboratory Grade
Application : Used in organic synthesis and pharmaceuticals
Oxoglutaric acid
Minimum Order Quantity : 500 Milliliters
Purity(%) : 99.99
Type : Industrial Lab Chemicals
Grade : Industrial
Application : Lab Chemicals
HP-53 Liquid crystal standard
Minimum Order Quantity : 500 Milliliters
Purity(%) : 99.99
Type : Industrial Lab Chemicals
Grade : Industrial
Application : Lab Chemicals
"Only deals in retail accepting orders upto 500ml only".
 |
ALPHA CHEMIKA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese


 Send Inquiry
Send Inquiry