Methyl thiocyanate
Methyl thiocyanate Specification
- Physical State
- Liquid
- Usage
- Laboratory reagent, chemical research
- Boiling point
- 130-132C
- Molecular Weight
- 73.12 g/mol
- Flash Point
- 34C
- Density
- 0.97 Gram per cubic centimeter(g/cm3)
- Purity
- 96%
- Packaging Type
- Bottle, Drum
- Molecular Formula
- C2H3NS
- Shelf Life
- 2 years
- Storage Instructions
- Store in a cool, dry, well-ventilated area away from heat and sources of ignition
- Size
- Varies (custom packaging available)
- Melting Point
- -29C
- CAS No
- 556-64-9
- Grade
- AR
- Type
- Industrial Lab Chemicals
- Application
- Pharmaceutical intermediates, Chemical synthesis
- Appearance
- Clear colorless to pale yellow liquid
- Purity(%)
- 96%
- UN Number
- UN3286
- Stability
- Stable under recommended storage conditions
- Solubility
- Slightly soluble in water, soluble in most organic solvents
- Odor
- Strong, unpleasant odor
- Synonyms
- Thiocyano methane, methylisothiocyanate
- Hazards
- Toxic by inhalation, ingestion, and skin absorption
- Refractive Index
- 1.486-1.488 (20°C)
Methyl thiocyanate Trade Information
- Minimum Order Quantity
- 500 Milliliters
- Supply Ability
- 50000 Milliliters Per Week
- Delivery Time
- 4-5 Days
About Methyl thiocyanate
Key Features and Benefits
Methyl thiocyanate offers high purity (96%) and is stable under standard storage conditions, ensuring reliable results in laboratory and industrial settings. Its versatility in application, particularly in the synthesis of pharmaceuticals and fine chemicals, makes it a valuable reagent. The chemical's clear to pale yellow appearance aids in ease of visual inspection during use.
Safe Handling and Storage Guidelines
Due to its toxicity and strong odor, methyl thiocyanate must be stored in a cool, dry, and well-ventilated space, away from heat and ignition sources. Proper packaging, typically in bottles or drums, ensures safety during storage and handling. Adhering to recommended storage conditions preserves both the stability and shelf life of the product, which is typically two years.
FAQ's of Methyl thiocyanate:
Q: How should methyl thiocyanate be safely handled in the laboratory?
A: Methyl thiocyanate must be handled with appropriate safety precautions, including the use of gloves, eye protection, and working within a well-ventilated fume hood. Avoid inhalation, ingestion, and skin contact due to its toxic nature.Q: What is the recommended storage process for methyl thiocyanate?
A: Store methyl thiocyanate in tightly sealed bottles or drums, placed in a cool, dry, and well-ventilated area. Keep it away from heat sources, direct sunlight, and ignition sources to maintain chemical stability and ensure safety.Q: Where is methyl thiocyanate commonly used?
A: It is commonly used in pharmaceutical research as an intermediate, as well as in chemical synthesis and laboratory applications. Its high purity and chemical stability make it suitable for industrial laboratory environments.Q: What are the hazardous properties of methyl thiocyanate, and how can exposure be minimized?
A: Methyl thiocyanate is toxic if inhaled, ingested, or absorbed through the skin, and emits a strong, unpleasant odor. Strict adherence to personal protective equipment guidelines and proper ventilation is crucial to minimize exposure risks.Q: When does methyl thiocyanate require replacement or disposal?
A: Replace or dispose of methyl thiocyanate if it has surpassed its two-year shelf life, if contamination is suspected, or if the container shows any damage or leakage. Always follow institutional and regulatory protocols for disposal.Q: What is the benefit of using methyl thiocyanate with 96% purity in chemical research?
A: Its high purity ensures consistent and accurate experimental outcomes, reducing impurities that might interfere with synthesis or analysis processes, thus enhancing the reliability of research results.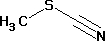

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Laboratory Chemicals Category
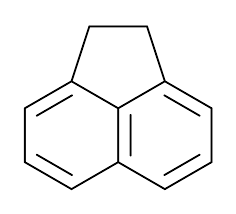
ACENAPHTHENE
Price 505 INR
Minimum Order Quantity : 100 Grams
Molecular Weight : 154.21 g/mol
Appearance : White to offwhite crystalline solid
Density : 1.06 Gram per cubic centimeter(g/cm3)
Usage : Used as an intermediate in organic synthesis
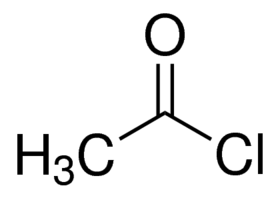
ACETYL CHLORIDE AR
Price 1300 INR
Minimum Order Quantity : 500 Milliliters
Molecular Weight : 78.5 g/mol
Appearance : Clear colourless to slightly yellow liquid
Density : 1.104 Gram per cubic centimeter(g/cm3)
Usage : For laboratory and industrial use only
ALUMINIUM OXIDE (active) BASIC
Price 475 INR
Minimum Order Quantity : 500 Grams
Molecular Weight : 101.96 g/mol
Appearance : Fine White Powder
Density : 3.97 Gram per cubic centimeter(g/cm3)
Usage : Used for adsorption and purification in chromatography, catalyst support, laboratory chemical
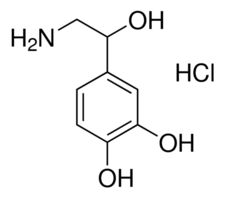
DL-nor-ADRENALINE (purified)
Price 22695.00 INR
Minimum Order Quantity : 1 Kilograms
Molecular Weight : 169.18 g/mol
Appearance : White to Offwhite Solid
Density : 1.3 Gram per cubic centimeter(g/cm3)
Usage : For laboratory use only
"Only deals in retail accepting orders upto 500ml only".
 |
ALPHA CHEMIKA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese


 Send Inquiry
Send Inquiry