Mercury (II) Thiocyanate
Price 35603 INR/ Gram
Mercury (II) Thiocyanate Specification
- Packaging Type
- Sealed plastic or glass containers
- Shelf Life
- 36 months if stored properly
- Molecular Weight
- 232.65 g/mol
- Molecular Formula
- Hg(SCN)2
- Purity
- 97%
- Physical State
- Solid
- Storage Instructions
- Store in a cool, dry, and well-ventilated place. Keep container tightly closed.
- Usage
- For chemical demonstrations such as Pharaohs Serpent, research, and analysis
- Melting Point
- 165 C (decomposes)
- Boiling point
- Not Applicable (decomposes before boiling)
- Size
- Custom packaging sizes available
- Density
- 3.268 Gram per cubic centimeter(g/cm3)
- CAS No
- 592-85-8
- Grade
- Laboratory Grade
- Type
- University Lab Chemicals
- Application
- Laboratory, Research, Educational Demonstrations
- Appearance
- White to grey crystalline powder
- Purity(%)
- 97%
- Decomposition
- Decomposes on heating to emit toxic fumes of mercury and sulfur oxides
- Hazard Class
- Toxic (H301; H331; H373); Environmentally hazardous substance
- UN Number
- UN 1641
- Odor
- Odorless
- Solubility
- Slightly soluble in water, soluble in ethanol and acetone
- EC Number
- 209-775-4
- Reactivity
- May react with oxidizers and acids
- Stability
- Stable under normal temperatures and pressures
- HS Code
- 2852
Mercury (II) Thiocyanate Trade Information
- Minimum Order Quantity
- 500 Grams
- Supply Ability
- 50000 Grams Per Week
- Delivery Time
- 4-5 Days
About Mercury (II) Thiocyanate
Owing to our rich knowledge as well as professional workforce, we have achieved a noted position as a reliable organization engaged in offering Mercury(II) Thiocyanate. Offered chemical is used in the preparation of other mercury or chemical compounds. It is used in mercuration reaction for chemistry and scientific laboratory. We use premium quality compounds, sourced from trusted vendors to process this chemical. Mercury Thiocyanate is widely used in chemical, pharmaceutical and bio chemistry. Clients can avail our chemical in different quantity packaging options.
Features:
Colorless white crystals
Longer shelf life
Soluble in water
LC3648 Mercury(II) thiocyanate GR for analysis Reag. Ph Eur
Order number | Packaging | Quantity | Price |
AC30648 | Glass bottle | 50 g | 26.55 |
AC30648 | Glass bottle | 250 g | 65.70 |
Product information | |
Grade | Reag. Ph Eur |
Synonyms | Mercury(II) rhodanide, Mercury(II) thiocyanate |
Hill Formula | C2HgN2S2 |
Chemical formula | Hg(SCN)2 |
HS Code | 2852 00 00 |
EC number | 209-773-0 |
Molar mass | 316.76 g/mol |
EC index number | 080-002-00-6 |
CAS number | 592-85-8 |
Chemical and physical data | |
Solubility | 0.7 g/l (25 C) |
Melting point | 165 C (decomposition) |
Molar mass | 316.76 g/mol |
Density | 3.71 g/cm3 (20 C) |
Bulk density | 750 kg/m3 |
pH value | ca.5 (50 g/l, H2O, 20 C) (slurry) |
Flash point | 120 C |
Safety information according to GHS | |
Hazard Statement(s) | H330: Fatal if inhaled. |
Precautionary Statement(s) | P273: Avoid release to the environment. |
Signal Word | Danger |
Hazard Pictogram(s) | |
Storage class | 6.1A Combustible substances, toxic |
WGK | WGK 3 highly water endangering |
Disposal | 20 |
Safety information | |
R Phrase | R 26/27/28-33-50/53-32 |
S Phrase | S 13-28-45-60-61 |
Categories of danger | very toxic, dangerous for the environment |
Hazard Symbol | Very toxic |
Transport information | |
Declaration (transport by sea) IMDG-Code | UN 1646 MERCURY THIOCYANATE, 6.1, II, Marine Pollutant: P, Segregation Group: 7 (Heavy metals and their salts (including their organometallic compounds)), 11 (Mercury and mercury compounds) |
Declaration (transport by air) IATA-DGR | UN 1646 MERCURY THIOCYANATE, 6.1, II |
Toxicological data | |
LD 50 oral | LD50 rat 46 mg/kg |
Specifications | |
Assay (complexometric) | 99.0 % |
Fe (Iron)hydrochloric acid | 0.002 % |
Substances not reducing by formic acid (residue on ignition) | 0.05 % |
Key Applications & Benefits
Mercury (II) Thiocyanate is widely recognized for its dramatic role in the Pharaoh's Serpent demonstration, offering unique visual insight into chemical decomposition. Its high purity (97%) and laboratory grade quality make it ideal for research and academic experiments. With custom packaging sizes available and a shelf life of up to 36 months, it is suitable for universities, research laboratories, and educational institutes requiring reliable and consistent reagents.
Safe Handling and Storage Guidelines
Due to its toxic properties, Mercury (II) Thiocyanate must be handled with strict safety measures. Store the product in tightly sealed plastic or glass containers, in a cool, dry, and well-ventilated area away from acids, oxidizers, and heat sources. Proper storage and adherence to safety guidelines are essential to maintain its stability, extend shelf life, and minimize health and environmental hazards.
FAQ's of Mercury (II) Thiocyanate:
Q: How should Mercury (II) Thiocyanate be safely stored and handled in a laboratory?
A: Store Mercury (II) Thiocyanate in tightly closed plastic or glass containers in a cool, dry, and well-ventilated area. Always avoid exposure to acids, oxidizers, or heat sources. Use personal protective equipment, such as gloves and safety goggles, to prevent contact due to its toxic nature.Q: What are the main uses and benefits of Mercury (II) Thiocyanate?
A: Mercury (II) Thiocyanate is primarily used for the Pharaoh's Serpent demonstration, showcasing thermal decomposition reactions, as well as for educational purposes, advanced research, and chemical analysis. Its high purity (97%) and stability make it suitable for controlled laboratory experiments.Q: When does Mercury (II) Thiocyanate decompose and what are the decomposition products?
A: Mercury (II) Thiocyanate decomposes when heated, typically around its melting point of 165C. The decomposition releases toxic fumes such as mercury vapors and sulfur oxides, making it crucial to conduct experiments under a fume hood or with adequate ventilation.Q: Where is Mercury (II) Thiocyanate typically applied or demonstrated?
A: This chemical is used in university and research laboratories, particularly for educational demonstrations of chemical reactions, as well as in research projects requiring a stable mercury compound. It is not intended for general or consumer use due to its toxicity.Q: What precautions should be taken when disposing of Mercury (II) Thiocyanate?
A: Dispose of Mercury (II) Thiocyanate as hazardous waste in accordance with local regulations. Avoid releasing it into the environment or through regular waste streams, as it is classified as an environmentally hazardous substance (UN 1641).Q: How does the solubility of Mercury (II) Thiocyanate affect its laboratory use?
A: Being only slightly soluble in water but readily soluble in ethanol and acetone, Mercury (II) Thiocyanate allows for selective solubility in various solvents, which may be advantageous in specific synthetic or analytical procedures.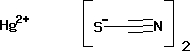

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Mercury Salts Category
Mercuric Oxide Red
Price 100-500 INR / Kilograms
Minimum Order Quantity : 25GM Kilograms
Physical State : Powder
Ph Level : Neutral
Appearance : Reddish orange powder
Type : Other
Mercuric Nitrate
Price 100-500 INR / Kilograms
Minimum Order Quantity : 25 Kilograms
Physical State : Solid
Ph Level : Acidic
Appearance : White Crystalline Powder
Type : Other
MERCUROUS CHLORIDE (anhydrous)
Price 31786 INR / Gram
Minimum Order Quantity : 500 Grams
Physical State : Solid
Ph Level : Neutral
Appearance : White crystalline powder
Type : Other, Anhydrous
MERCURIC SULPHATE AR
Price 3057 INR / Gram
Minimum Order Quantity : 500 Grams
Physical State : Solid
Ph Level : Acidic in water
Appearance : A white crystalline powder
Type : Other, Inorganic compound
"Only deals in retail accepting orders upto 500ml only".
 |
ALPHA CHEMIKA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese


 Send Inquiry
Send Inquiry






